Parylene C Biocompatibility: Properties, Safety, and Medical Use Cases
Overview & Key Takeaways
Parylene C is biocompatible and meets USP Class VI and ISO 10993 standards — the two primary benchmarks for medical device materials. It is the most widely deployed variant in the Parylene family for medical and implantable device applications. Its chlorinated para-xylylene structure delivers superior moisture barrier performance and chemical resistance while maintaining full biocompatibility with human tissue and fluids. Engineers evaluating Parylene C should weigh its exceptional protective properties against its mechanical limitations relative to bulk structural materials.
- USP Class VI and ISO 10993 compliance: Parylene C meets the two primary biocompatibility standards for medical devices, including passing cytotoxicity, sensitization, genotoxicity, and implantation tests. Independent third-party testing confirms it causes no measurable harm to tissues or body fluids.
- Chemical inertness: Parylene C does not leach plasticizers, solvents, or catalysts into surrounding tissue. Deposited as a pure polymer with no additives, it resists bodily fluids, enzymes, and varying pH levels without degradation over time.
- PFAS-free formulation: Unlike Teflon (PTFE), Parylene C contains no per- or polyfluoroalkyl substances and is both REACH and RoHS compliant — a meaningful differentiator for manufacturers navigating tightening PFAS regulations.
- Pinhole-free conformal barrier: Chemical vapor deposition produces a continuous, void-free coating at thicknesses as low as 1 micron, conforming to complex geometries and fine features while fully isolating device surfaces from biological environments.
- High dielectric strength: Parylene C achieves up to 7,000 V/mil, providing reliable electrical insulation for implantable electronics, sensors, and active devices operating in the body.
- Tradeoff vs. structural materials: Parylene C is not suitable as a load-bearing or structural material — its tensile and yield strength are lower than metals and ceramics. Applications requiring mechanical strength must pair Parylene with a rigid substrate. CVD application also requires specialized equipment and process integration not needed for simpler liquid coating methods.
Parylene C is widely considered one of the most biocompatible conformal coatings used in medical devices. It forms a chemically inert, pinhole-free barrier that protects both the device and the human body from harmful interactions.
Because of its stability, low permeability, and proven safety profile, Parylene C is commonly used in implants, surgical tools, and wearable medical electronics.
What Does "Biocompatibility" Mean for Parylene C?
Biocompatibility refers to a material's ability to perform safely in contact with the human body without causing adverse reactions such as toxicity, inflammation, or immune response.
Parylene C achieves this by acting as a non-reactive, protective barrier between device materials and biological environments. It prevents leaching, corrosion, and chemical interaction — key risks in medical applications.
What Biocompatibility Standards Does Parylene C Meet?
Parylene C satisfies both primary biocompatibility standards used by medical device manufacturers and regulatory bodies:
- USP Class VI — the highest classification in the U.S. Pharmacopeia's plastics testing protocol, requiring passing results for systemic injection, intracutaneous injection, and implantation tests
- ISO 10993 — the international standard series for biological evaluation of medical devices, covering cytotoxicity, sensitization, and genotoxicity, among others
Independent testing confirms Parylene C passes both standards. Its chemical inertness — meaning it does not react with bodily fluids, enzymes, or varying pH environments — is the primary reason for its compliance.
Why Is Parylene C Considered Biocompatible?
- Chemically inert: Does not react with bodily fluids or tissues
- Pinhole-free coating: Provides complete, uniform coverage with no gaps or voids
- Low permeability: Blocks moisture, gases, and contaminants
- Non-toxic barrier: Prevents harmful substances from leaching into the body
- PFAS-free: Contains no per- or polyfluoroalkyl substances — REACH and RoHS compliant
Parylene C's ability to create a continuous, defect-free film is critical. Unlike liquid coatings, it forms a uniform layer even on complex geometries, reducing exposure risks.
Key Properties That Enable Biocompatibility
1. Exceptional Barrier Protection
Parylene C has extremely low permeability to moisture and gases, which helps prevent corrosion and contamination in harsh biological environments.
2. Chemical Resistance
It resists a wide range of chemicals, ensuring long-term stability when exposed to bodily fluids, enzymes, and varying pH levels.
3. Electrical Insulation
Parylene C has a dielectric strength of up to 7,000 V/mil, making it ideal for implantable electronics and sensors that must function reliably in the body.
4. Conformal Coverage
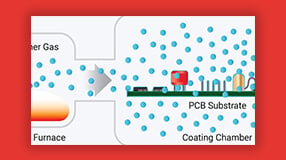
The chemical vapor deposition (CVD) process allows Parylene to coat intricate surfaces evenly at thicknesses as low as 1 micron, eliminating gaps that could compromise safety or device performance.
How Does Parylene C Compare to Other Medical Coating Materials?
| Property | Parylene C | Silicone | Epoxy | Teflon (PTFE) |
|---|---|---|---|---|
| Biocompatibility Standard | USP Class VI, ISO 10993 | ISO 10993 | Moderate | ISO 10993 |
| Moisture Barrier | Excellent | Moderate | Good | Good |
| Conformality | Complete coverage | Limited | Limited | No |
| Thickness Control | Precise (micron-level) | Variable | Variable | 12–25 µm |
| PFAS-Free | Yes | Yes | Yes | No |
| Dielectric Strength | Up to 7,000 V/mil | ~500 V/mil | ~400 V/mil | ~2,000 V/mil |
This combination of properties is why Parylene C is often selected for critical medical and implantable applications — particularly where Teflon alternatives are needed due to tightening PFAS regulations.
Common Medical Applications of Parylene C
- Implantable devices (pacemakers, stents, cochlear implants)
- Cardiac assist devices — reduces thrombogenicity without altering device geometry
- Needles and cannulas — reduces friction for easier insertion and improved patient comfort
- Wearable medical electronics
- Surgical tools and instruments
- Microelectromechanical systems (MEMS)
- Drug delivery devices
The global Parylene market was valued at $98.38 million in 2023 and is projected to grow to $118.52 million by 2030, driven in large part by expanded medical device applications.
Does Parylene C Improve Device Safety?
Yes. Parylene C enhances safety in two critical ways:
- Protects the body: Prevents exposure to device materials and potential toxins by creating a chemically inert barrier between the substrate and surrounding tissue
- Protects the device: Shields electronics from moisture, corrosion, and biological degradation — extending functional lifespan in demanding in-vivo environments
What Are the Limitations of Parylene C in Medical Applications?
While Parylene C is highly biocompatible, it is not universally ideal for every application:
- Mechanical load-bearing: Parylene's tensile strength is lower than metals and ceramics — it is not appropriate for structural applications without a rigid substrate
- Adhesion on certain substrates: Some materials require surface preparation or adhesion promoters before coating
- Application process complexity: CVD requires specialized equipment and batch processing, unlike simpler dip or spray coating methods
These challenges are typically addressed through proper engineering and process optimization — and in most cases, Parylene's performance advantages justify the added process complexity.
FAQ: Parylene C Biocompatibility
Is Parylene C safe for implantable medical devices?
Yes. Parylene C meets USP Class VI and ISO 10993 biocompatibility standards — the primary requirements for implantable and body-contact medical devices. It is non-toxic, physiologically non-reactive, and has a decades-long track record in implantable applications, including pacemakers, stents, and cochlear implants.
Is Parylene FDA approved?
Parylene itself is not an FDA-approved material — the FDA approves devices, not coating materials in isolation. However, Parylene C is widely used in devices that have received FDA clearance or approval, and its USP Class VI and ISO 10993 compliance makes it well-suited for inclusion in FDA submissions.
Does Parylene C prevent corrosion?
Yes. Its low permeability, pinhole-free structure, and chemical inertness make Parylene C highly effective at preventing corrosion caused by moisture, bodily fluids, and enzymatic activity.
Is Parylene PFAS-free?
Yes. Parylene C contains no per- or polyfluoroalkyl substances (PFAS) and is both REACH and RoHS compliant — unlike Teflon (PTFE), which is classified as a PFAS material. This makes Parylene a strong alternative for manufacturers navigating tightening PFAS regulations.
How does Parylene compare to silicone coatings?
Parylene offers better moisture resistance, more precise thickness control, and more uniform coverage — especially on complex geometries. Silicone coatings are applied at significantly greater thickness and cannot achieve the pinhole-free conformality Parylene delivers via CVD.
How is Parylene applied to medical devices?
Parylene is deposited via chemical vapor deposition (CVD) — a room-temperature process that conformally coats all exposed surfaces regardless of geometry. The process leaves no liquid residue and requires no curing step.
Read about how to incorporate Parylene into your process
Learn More About Parylene Coatings
If you're evaluating Parylene for medical or high-reliability applications, understanding its advantages and limitations is critical.
Explore more about Parylene coating properties or learn how it compares to other materials in our Parylene vs. liquid coatings guide.
Learn more in our webinar, Parylene: Pros, Cons, and Practicalities
An Overview of Our Parylene Coating Equipment
Parylene Coating Removal - Methods & Best Practices for Each
Parylene Conformal Coatings - Parylene Conformality Explained
Mallory is a veteran writer with over a decade of writing experience and has spent over five years at HZO learning about coating technology from the best minds in the industry. Professionally, Mallory is especially interested in the process of problem-solving and watching how the engineering team develops solutions that ensure business requirements are met. Over her years at HZO, Mallory's writing has been cited in industry whitepapers, including "Parabolic Model for Optimum Dry Film Thickness (DFT) of Corrosion Protective Coatings" and "Universal Approach to Integrating Reduced Graphene Oxide into Polymer Electronics." All of Mallory’s blogs are reviewed for accuracy before publication.
Additional Resources
Learn About Parylene Properties With Our Resource Page
Learn How To Incorporate Conformal Coating Into Your Manufacturing Process

Learn About the Thin Film Coating Properties and Processes In Our Webinar

Learn How to Bring Parylene Deposition In-House